Recalls and alerts more than 4 years old are automatically archived. While this information can still be accessed in the database, it has not been altered or updated since it was archived. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats by contacting us.
Unauthorized Botox and other health products seized from Arshia Hair Salon and Spa in Toronto May Pose Serious Health Risks
- Starting date:
- May 19, 2018
- Type of communication:
- Advisory
- Subcategory:
- Drugs
- Source of recall:
- Health Canada
- Issue:
- Unauthorized products
- Identification number:
- RA-66830
- Issue
- Products affected
- What you should do
- Who is affected
- Background
- Media enquiries
- Public enquiries
- What Health Canada is doing
- Images
Issue
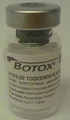
Health Canada is advising Canadians that it has seized an unauthorized “Botox” product and other unauthorized health products from Arshia Hair Salon and Spa (6062A Yonge Street) in Toronto, ON, because they may pose serious health risks. These products may have been administered as injectable treatments for cosmetic purposes.
Unauthorized health products have not been approved by Health Canada, which means that they have not been assessed for safety, effectiveness and quality. Selling unauthorized health products in Canada is illegal.
Products affected
- Botox botulinum toxin type A (a version that is not authorized in Canada)
- Hylase
- Mesoline Bodyfirm
- Mesoline Slim
- Mesoline Tight
What you should do
- Do not use these unauthorized products from Arshia Hair Salon and Spa, or anywhere else. Consult your health care professional if you have used or have been administered these products and have health concerns.
- Read product labels to verify that health products have been authorized for sale by Health Canada. Authorized health products have an eight-digit Drug Identification Number (DIN), Natural Product Number (NPN) or Homeopathic Drug Number (DIN-HM). You can also check whether products have been authorized for sale by searching Health Canada's Drug Product Database and Licensed Natural Health Product Database or the Medical Devices Active Licence Listing Database.
- Report adverse events to health products to Health Canada by calling toll-free at 1‑866‑234‑2345, or by reporting online, by mail or by fax.
- Report complaints about health products to Health Canada by calling toll-free at 1‑800‑267‑9675, or complete an online complaint form.
Who is affected
Consumers who have been administered or used any of these unauthorized health products.
Background
Botulinum toxin type A is used to treat severe muscle spasms in the neck, eye and foot, as well as chronic migraines, urinary incontinence, and excessive sweating. It is also used for cosmetic purposes to treat facial wrinkling. Authorized botulinum toxin type A products should be used only under specialist supervision and only if the benefits of treatment are considered to outweigh the risks. Potential risks associated with injecting an unauthorized Botulinum toxin type A product can range from mild local paralysis to death. All products administered by injection in Canada must be authorized for sale by Health Canada.
Unauthorized injectable health products for cosmetic purposes carry significant risk because of the potential for infection, scarring and poor outcomes. These products have not been assessed for safety, effectiveness or quality.
Related links
Stay connected with Health Canada and receive the latest advisories and product recalls.
Media enquiries
Health Canada
(613) 957-2983
Public enquiries
(613) 957-2991
1-866 225-0709
What Health Canada is doing
Health Canada seized the unauthorized health products. Should additional retailers or distributors be identified, Health Canada will take appropriate action and inform Canadians as necessary.
Images
Select thumbnail to enlarge - opens in a new window
- Date modified: